Introduction: The branch of mineralogy dealing with the study of crystals is called
Crystallography. It deals with the study of the laws that govern their growth, external shape and internal structure.
Crystal: “ A homogenous solid bounded by naturally formed plane surfaces termed faces, which can be related to a regular arrangement of atoms” (Gribble, 1991).
The following characteristics are present in crystals:
- A crystal has a regular polyhedral shape with faces, or flat surfaces.
2. it has a characteristic interior atomic structure; and it can only develop its regular geometry under the right physicochemical conditions. Slow cooling produces well-developed crystals.
3. A crystal is considered perfect if it has both exterior form and internal atomic structure. However, something is considered crystalline if only the internal atomic structure is present and the appropriate outward shape has not developed. A substance is said to be amorphous if it lacks both external form and internal atomic structure.

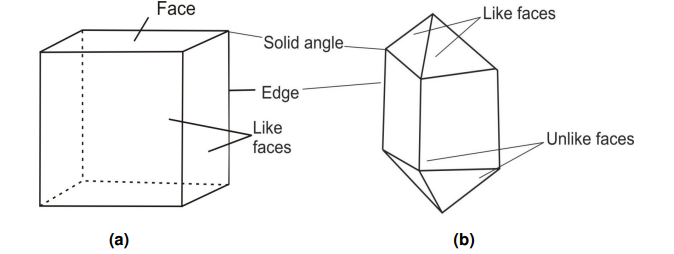
- Faces: The flat surfaces that form the outer sides of a crystal are called faces. These faces are parallel to the internal layers, or net planes, of the crystal structure. Usually, the faces are flat, but in some minerals—like siderite, dolomite, and diamond—they can be slightly curved.
If all the faces of a crystal look the same, the crystal is said to have like faces (for example, galena). But if the faces are of different shapes or types, the crystal has unlike faces (for example, zircon).
2. Edges: are formed where two adjacent faces of a crystal meet. Each edge is naturally straight, and its position depends entirely on how the adjoining faces are arranged.
3. Solid angles: They are formed at the intersection of three or more than three faces.
Solid angle is also known as a vertex. The relationship
between these elements in a crystal can be expressed by the formula,
f + c = e + 2
where,
f- number of faces
c-number of solid angles
e- number of edges
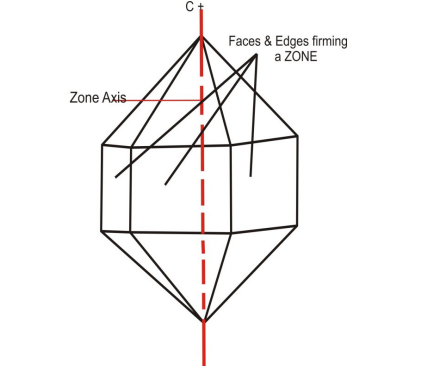
4. Zone and Zone Axis: The study of crystals shows that in many cases, the faces are arranged so that the edges formed by their intersections run parallel to each other. Each group of such parallel edges corresponds to a specific set of faces. A zone is defined as a group of faces whose intersections produce these parallel edges. An imaginary line inside the crystal, parallel to all the edges in a zone, is called the zone axis.